I belong to the Institute of Biological and Organic Chemistry, University of Fukui. Do you know the light? We can’t live without light. Therefore light is important to us. Our laboratory conducts research using this light.
Organic chemistry is one of chemistry. Chemical substances are made by organic synthesis. There are various kinds of organic synthesis. Most organic synthesis requires high temperatures and metals, but such reactions are not environmentally friendly. On the other hand, the reaction using light is easy to experiment and environmentally friendly. We can conduct organic synthesis easily and environmentally by using photoreaction.
Two kinds of light are used for photoreaction. It is ultraviolet light and visible light. We use ultraviolet light in our laboratory. High pressure mercury lamp is used for ultraviolet light irradiation.
There is a big reason for the reaction to progress as light hits. The reason is that chemical substances become excited state with high energy due to light absorption. This excited state has an electron transfer effect on other substances. This effect becomes the starting point of chemical reaction.
Since the reaction after this will be content that is rather difficult, I will omit it.
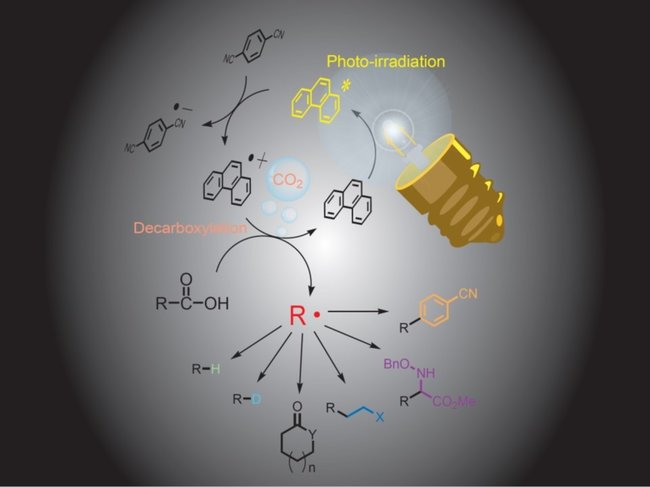
I will introduce the past research in which this reaction was used. One of them is decarboxylation. The decarboxylation reaction which has been carried out using dangerous metal up to now becomes possible by photoreaction. When this reaction occurs, a compound called a carboxylic acid (a compound having -COOH) turns into a highly reactive compound called radical(→R・). The reaction progresses as this radical compound acts on other substances.
The research I am doing is different from this class. My research is multipurpose coupling reaction, not decarboxylation. I will introduce this coupling reaction.
In my research as well, light irradiation is used, and also molecules for getting excited are necessary. My research differs from the decarboxylation reaction in that the compound used is an unsaturated hydrocarbon compound rather than a carboxylic acid. By using a reaction on this compound it turns into a substance called a radical cation(→R・+). Since radical cation has two parts to react and react, it can react with two molecules. This allows three molecules to be coupled at once by photoreaction. This reaction is a new reaction that has yet to be achieved by anyone worldwide!
Organic chemistry is one of chemistry. Chemical substances are made by organic synthesis. There are various kinds of organic synthesis. Most organic synthesis requires high temperatures and metals, but such reactions are not environmentally friendly. On the other hand, the reaction using light is easy to experiment and environmentally friendly. We can conduct organic synthesis easily and environmentally by using photoreaction.
Two kinds of light are used for photoreaction. It is ultraviolet light and visible light. We use ultraviolet light in our laboratory. High pressure mercury lamp is used for ultraviolet light irradiation.
There is a big reason for the reaction to progress as light hits. The reason is that chemical substances become excited state with high energy due to light absorption. This excited state has an electron transfer effect on other substances. This effect becomes the starting point of chemical reaction.
Since the reaction after this will be content that is rather difficult, I will omit it.
I will introduce the past research in which this reaction was used. One of them is decarboxylation. The decarboxylation reaction which has been carried out using dangerous metal up to now becomes possible by photoreaction. When this reaction occurs, a compound called a carboxylic acid (a compound having -COOH) turns into a highly reactive compound called radical(→R・). The reaction progresses as this radical compound acts on other substances.
The research I am doing is different from this class. My research is multipurpose coupling reaction, not decarboxylation. I will introduce this coupling reaction.
In my research as well, light irradiation is used, and also molecules for getting excited are necessary. My research differs from the decarboxylation reaction in that the compound used is an unsaturated hydrocarbon compound rather than a carboxylic acid. By using a reaction on this compound it turns into a substance called a radical cation(→R・+). Since radical cation has two parts to react and react, it can react with two molecules. This allows three molecules to be coupled at once by photoreaction. This reaction is a new reaction that has yet to be achieved by anyone worldwide!
Our research; photo decarboxylation